After two publications in July 2002 informed of negative side effects associated with estrogen/progestin combination therapy, use of previous steadily rising Hormone Replacement Therapy from 1995 - 2002 suddenly declined in 2003. Data was observed through two separate national databases, from the years 1995-2003 to conduct this analysis.
The reason for the steady rise of hormone therapy among women was due to a lack of definitive information concerning the risks and benefits of usage combined with clever media tactics and successful marketing strategies. The halt in progress came when the Heart and Estrogen/Progestin Replacement Study (HERS) and Women's Health Initiative (WHI) both conducted studies to determine whether or not hormone combination estrogen/progestin use really offered cardiovascular benefits as was suggested.
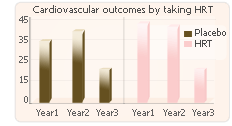
The outcome of both served to disprove said theory. The HERS testing concluded that post-menopause hormone therapy with combination estrogen/progestin failed to offer any benefit to women with cardiovascular disease. The WHI trial demonstrated that hormone therapy with an estrogen/progestin combination caused an increased risk of breast cancer and cardiovascular disease in women who have surpassed menopause.
In 2001 hormone therapy patients were tested, finding 14,500 adverse issues attributed to oral estrogen plus progestin. Of the women tested, 3,500 had cardiovascular disease, 2000 had breast cancer, 4000 had strokes and 5000 reported a case of pulmonary embolism.
The databases that conducted these surveys were the National Disease and Therapeutic Index (NDTI) database and the National Prescription Audit (NPA) database. The NDTI offered information on patient visits to physicians and associated medication use. Physicians described all diagnoses and indicated newly prescribed and continuing medications used specifically for each diagnosis. The NPA gave data based on the number of hormone therapy prescriptions filled at approximately 20,000 retail pharmacies, independent pharmacies, mail order pharmacies and mass merchandise and discount houses. Data was given for all hormone therapy formulations and individuals brands. Such included, oral, transdermal, vaginal applications with combined estrogen/progestin. Both data bases gave information from 1995 to 2003. The results from both databases were combined to estimate the number of women receiving hormone therapy in the form of estrogen and progestin combination.
The conclusive results combined from both databases showed a significant drop in the use of hormone therapy prescriptions after the publications made by WHI and HERS in July 2002. Based on data from July 2003, hormone therapy prescriptions declined by 38 percent.
In 2003, the latest guidelines suggested against routine hormone therapy use for chronic conditions and that current users lowered doses toward discontinuation. If a woman's vasomotor symptoms returned after discontinued use, alternative treatment were suggested, including lower dose hormone therapy or herbal products.
The continuing decline in hormone therapy treatment is most likely in response to the side effects associated with them. Specifically, women shy away from estrogen/progestin combinations to avoid cardiovascular disease and breast cancer risk. Physicians predict the number of hormone therapy prescriptions to steadily decline due to such factors.