After menopause, the level of estrogen in a women's body is significantly low. Because of the natural loss of estrogen, postmenopausal women experience a slew of undesirable side affects; chiefly, osteoporosis. To treat and prevent osteoporosis, hormone replacement therapy (HRT) has been used for decades. Recently, the Food and Drug Administration (FDA) has approved two new drugs, alendronate sodium and raloxifene hydrochloride, as an alternative for HRT and estrogen replacement for osteoporosis. Since data is limited on these new drugs, a study was conducted to determine their efficacy and side effects in comparison to HRT for osteoporosis. The risks on hip fracture, coronary heart disease (CHD), breast cancer and life expectancy were also compared for the three treatment methods.
It is not uncommon for postmenopausal women to worry about the life long effects of estrogen deficiency. Widely used HRT, estrogen combined with progestin, prevents accelerated bone loss that occurs after menopause and may even decrease the risk of developing CHD. It may however increase the risk of breast and endometrial cancers. The new therapies, alendronate sodium, and raloxifene hydrochloride, have been studied as new sources for estrogen replacement and have proven to decrease the risk of verbal fractures but without the same cancer risks caused by HRT.
Both however, also lack some of the benefits associated with HRT. Raloxifene, a nonsteroidal compound with mixed estrogenic and antiestrogenic properties, does not increase the risk of endometrial cancer, may decrease the risk of breast cancer and can lower serum total cholesterol levels. Alendronate, a bisphoshonate, may inhibit bone resorption and has no known impact on CHD or either cancers associated with HRT.
For over 30 years, postmenopausal women taking HRT have experienced a decrease in fracture rates, the incidence of CHD and an increase in survival rates. Because they are new, raloxifene and alendronate therapy could only go through limited measures of clinical efficacy. Each will require decades of follow-up studies to determine a more accurate measure of impact on hip fracture and CHD.
The study was done on healthy, white 50 year old postmenopausal women who were receiving one of the three treatments for estrogen deficiency.
Effects of HRT
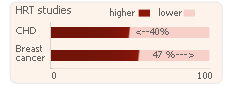
Studies suggest that HRT:
- Lowers the risk of developing CHD by 40%
- Increases the risk of breast cancer by 47%
- Shows no impact (when used with progestin) on endometrial cancer
Effects of Raloxifene Therapy
Long term impact of raloxifene therapy on breast cancer cannot yet be established. During the three years of randomized studies of 10,385 postmenopausal women taking raloxifene there was an increase of 38% for breast cancer. This study however assumes that raloxifene therapy only lowers the risk of breast cancer during the first five years of treatment, rather than preventing it completely.
Impact of Life Expectancy
HRT increased life expectancy among postmenopausal women more than raloxifene and alendronate therapy. It is important to note that life expectancy gains depended on the individual woman's risk profile thus no treatment was proved better or worse for life expectancy. However, women at a high risk for CHD should take HRT while women at high risk for breast cancer would gain from taking raloxifene.
Impact on Hip Fracture
All three therapies showed similar efficacies in preventing hip fractures.
Each individual woman must decide what the best treatment for estrogen replacement is for her after menopause. Her health and risk factors must come into play when considering using HRT or other newer forms of estrogen replacements such as raloxifene and alendronate therapy as none are without side effects.