What is Prenatal Genetic Testing?
Prenatal genetic testing is a set of tests that are administered during pregnancy to determine whether the fetus is at risk of or has certain genetic disorders.
A couple in which one or both partners are carriers of a certain genetic disorder often undergo preconception genetic testing, also known as carrier screening, to assess the risk of inheritance before conceiving.
Who Needs Prenatal Genetic Testing?
While all women are offered to undergo non-invasive prenatal genetic screening, diagnostic tests are typically recommended when the screening tests have shown positive or inconclusive results or women are at an increased risk for genetic disorders, including:
- Advanced maternal age
- Chronic maternal conditions, like high blood pressure, lupus, epilepsy, or diabetes
- History of miscarriages or having a child with birth defects
- Family history of genetic disorders
- Being a carrier of or having a partner who is carrier of a genetic disorder
- Multiple gestation
- Certain ethnicity associated with a higher risk of some genetic disorders
Which Disorders Do Prenatal Genetic Tests Detect?
Prenatal genetic tests can detect chromosome abnormalities, gene mutations, and neural tube defects that would result in birth defects and genetic disorders, including the following:
- Down syndrome
- Edwards syndrome
- Patau syndrome
- Cystic fibrosis
- Tay-Sachs disease
- Thalassemia
- Sickle cell anemia
- Fragile X syndrome
- Congenital heart defects
- Anencephaly
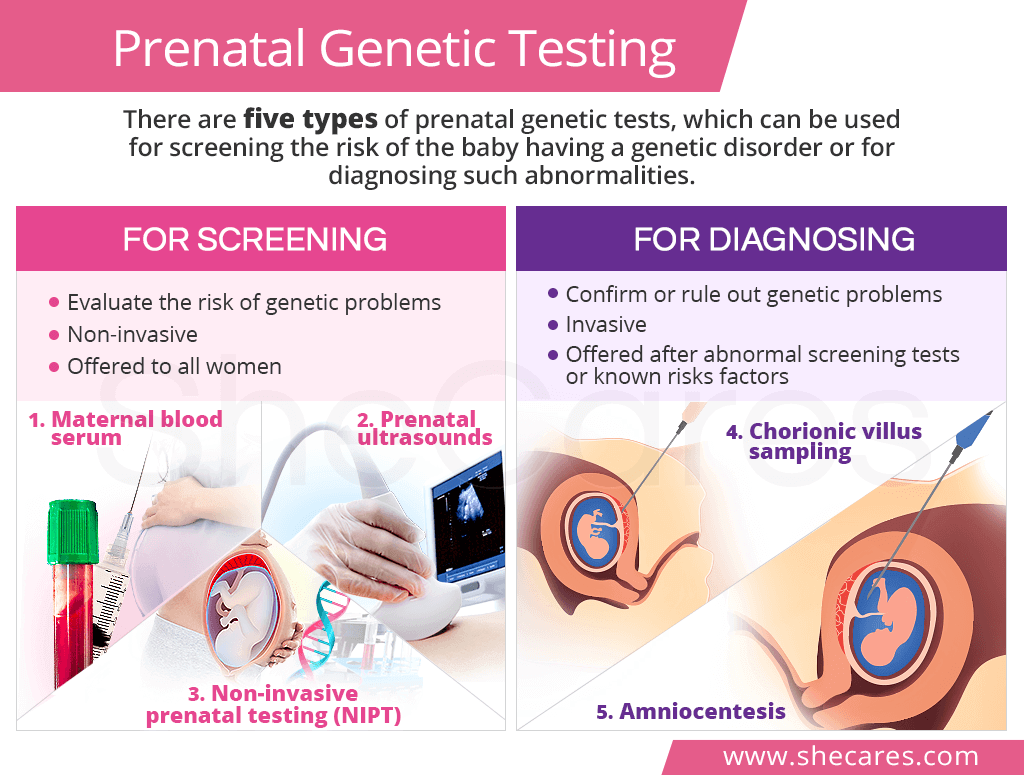
Types of Prenatal Genetic Testing
The five types of prenatal genetic testing are divided in two groups, the ones used for screening and others used for diagnosing genetic abnormalities.
For Screening:

The first group of tests is aimed at genetic screening during pregnancy to evaluate the risk of one's baby having a genetic abnormality in comparison to the general population. They are non-invasive, cause no serious side effects, and are offered to pregnant women first, unless there are risk factors or other indications that necessitate going straight to diagnostic tests.
Maternal Blood Serum
Maternal blood serum is a type of prenatal blood test, which is typically offered to all pregnant women in the first and second trimester to determine the risk for chromosomal abnormalities.
1st Trimester Maternal Blood Serum. Done between the 11th and 14th weeks of pregnancy, this type of prenatal genetic testing evaluates the risk for aneuploidy (missing or extra genes). The results are generally combined with nuchal translucency screening as part of the “first trimester screening. “
2nd Trimester Maternal Blood Serum (also called quad screen or multiple marker screen). Done between the 15th and 20th weeks of pregnancy, this fetal genetic testing screens for both aneuploidy and neural tube defects. The results are combined with an anomaly ultrasound as part of the “second trimester screening.”
Prenatal Ultrasounds
Prenatal ultrasounds are non-invasive examinations suggested to all pregnant women to monitor fetal development, detect pregnancy complications, and determine the risk of birth defects in the fetus.
1st Trimester Ultrasound: Nuchal Translucency Screening. Performed between the 11th and 14th weeks, it measures the thickness of the back of the baby's neck. This area appears thicker if there is a risk of chromosomal defects. The results are typically combined with the first trimester maternal blood serum.
2nd Trimester Ultrasound: Anomaly Ultrasound. Done between the 18th and 20th weeks, it checks for physical signs of potential congenital malformations in the baby's body, including the brain, heart, limbs, spine, and more. The results are typically combined with the second trimester maternal blood serum.
Non-Invasive Prenatal Testing (NIPT)
NIPT, which is also known as cell-free fetal DNA testing, is not offered to all women. Instead, it is recommended to those at an increased risk of having a baby with a genetic disorder, excluding low-risk and multiple gestation.
This innovative type of prenatal genetic testing is usually done around the 10th week of pregnancy. It uses a maternal blood sample to test small particles of DNA that are released into the bloodstream from the placenta.
For Diagnosing:
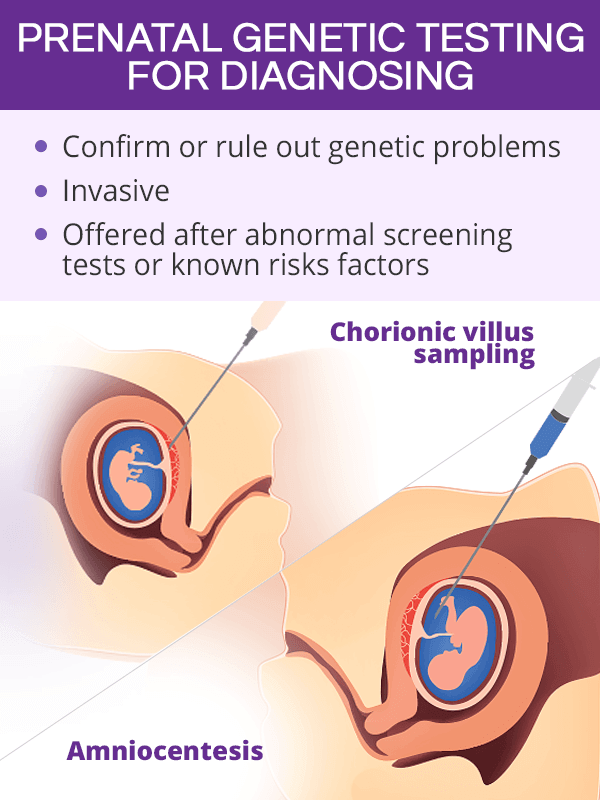
The second group of prenatal genetic tests are diagnostic tests. Being more invasive, they are offered to women when the results of the screening tests indicated some possible abnormalities or to women have known risk factors.
Chorionic Villus Sampling
Chorionic villus sampling (CVS) is a diagnostic genetic test during which a tissue from the placenta is withdrawn from the womb and examined for birth defects and genetic conditions, like cystic fibrosis or sickle cell anemia. CVS is done in the first trimester, between the 10th and 13th week of pregnancy.
Amniocentesis
Amniocentesis is another diagnostic prenatal genetic testing during which a sample of the amniotic fluid is withdrawn from the womb and tested for genetic disorders and birth defects. It is usually done in the second trimester, between the 15th and 20th weeks.
Because chorionic villus sampling and amniocentesis are invasive genetic prenatal tests, they carry a small risk of a miscarriage, about 1%1 and 0.25-0.5%2, respectively.
Key Takeaways
Though not mandatory, all five types of prenatal genetic testing are an important part of prenatal care as they can determine if the baby is at risk of or has certain genetic conditions. All women will be offered a non-invasive genetic screening during pregnancy, which consists of blood tests and ultrasounds. Some, including those with known risk factors like advanced age or chronic diseases, might be then recommended a more invasive diagnostic genetic testing, like amniocentesis.
However, the difficulty of this decision doesn't only lie in possible miscarriage risk involved with diagnostic genetic tests, but also with the question of what to do next. Some couples choose not to know ahead of time if their baby has a genetic disorder, while others might want to know to have time to prepare, arrange for adoption, or consider undergoing abortion. All in all, staying well-informed and doing research can help a couple make the best choices for their future.
Sources
- American College of Obstetrics and Gynecologists. (2019). Prenatal Genetic Diagnostic Tests. Retrieved July 9, 2019 from https://www.acog.org/Patients/FAQs/Prenatal-Genetic-Diagnostic-Tests
- American College of Obstetrics and Gynecologists. (2017). Prenatal Genetic Screening Tests. Retrieved July 9, 2019 from https://www.acog.org/Patients/FAQs/Prenatal-Genetic-Screening-Tests
- American Society of Human Genetics. (n.d.). Non-invasive Prenatal Genetic Testing (NIPT). Retrieved July 9, 2019 from https://www.ashg.org/education/pdf/NIPT-final.pdf
- Better Health Channel. (n.d.). Pregnancy tests - maternal serum screening. Retrieved July 9, 2019 from https://www.betterhealth.vic.gov.au/health/conditionsandtreatments/pregnancy-tests-maternal-serum-screening
- Genetics Education. (2018). Prenatal Testing Overview. Retrieved July 9, 2019 from https://www.genetics.edu.au/publications-and-resources/facts-sheets/fact-sheet-24-prenatal-testing-overview
- Medline Plus. (2015). Prenatal Testing. Retrieved July 9, 2019 from https://medlineplus.gov/prenataltesting.html
- Royal College of Obstetricians & Gynaecologists. (2011). Chorionic villus sampling and amniocentesis. Retrieved July 9, 2019 from https://www.rcog.org.uk/globalassets/documents/patients/patient-information-leaflets/pregnancy/pi-chorionic-villus-sampling-and-amniocentesis.pdf
Footnotes:
- American Pregnancy Association. (2016). Chorionic Villus Sampling (CVS). Retrieved July 9, 2019 from https://americanpregnancy.org/prenatal-testing/chorionic-villus-sampling/
- American Pregnancy Association. (2016). Amniocentesis. Retrieved July 9, 2019 from https://americanpregnancy.org/prenatal-testing/amniocentesis/
Further Reading:
- CDC. (2018). Genetic Counseling. Retrieved July 9, 2019 from https://www.cdc.gov/genomics/gtesting/genetic_counseling.htm
- Genetics Home Reference. (2019). What is genetic testing? Retrieved July 9, 2019 from https://ghr.nlm.nih.gov/primer/testing/genetictesting
- National Human Genome Research Institute. (2018). 15 for 15: Noninvasive Prenatal Genetic Testing. Retrieved July 9, 2019 from https://www.genome.gov/dna-day/15-for-15/noninvasive-prenatal-genetic-testing
- NIH Record. (2015). Prenatal Testing Revolutionizing Fetal Care. Retrieved July 9, 2019 from https://nihrecord.nih.gov/newsletters/2015/03_13_2015/story2.htm