This study aims to determine the safety and effectiveness of fluoride in increasing spinal bone mass and reducing spinal fractures in older women with established osteoporosis, who may be using hormone replacement therapy. A comparison was made between a combination of sustained-release sodium fluoride, calcium citrate, and cholecalciferol (SR-NaF group) and calcium and cholecalciferol alone (control group). Due to the fact that estrogen use is popularly used by postmenopausal women for numerous reasons, for example, as a hormone replacement therapy, current estrogen use was allowed but patients were specifically arranged to ensure equal distribution between groups.
Osteoporosis is a widespread condition affecting older generations, particularly postmenopausal women who may be using hormone replacement therapy. Bone turnover may also increase with aging. Currently, there are no official anabolic or bone-forming agents to treat osteoporosis. Such therapies influence bone mass through the stimulation of osteoblastic activity, resulting in increased bone mass. Sodium fluoride is known to stimulate bone formation and when given in low doses, bone mass is increased and risk for vertebral fractures is reduced in osteoporosis sufferers, including postmenopausal women who may be receiving hormone replacement therapy. However, the efficacy and safety of sodium fluoride is uncertain.
Analyses of patients with osteoporosis who may be using hormone replacement therapy treated with constant and/or high-dose sodium fluoride have shown that risk for vertebral fractures was unaffected compared with that of controls, despite increases in spinal bone mass in the sodium fluoride-treated group. Furthermore, increased numbers of nonvertebral fractures in sodium fluoride-treated patients lead to concern about impaired bone quality and reduced bone strength. Additionally, serious side effects have been associated with sodium fluoride.
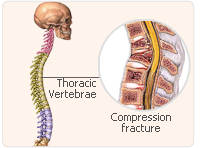
Findings from this study show that the combination therapy consisting of sustained-release sodium fluoride, calcium, and cholecalciferol in older women with established osteoporosis who may be using hormone replacement therapy significantly reduced the risk for subsequent vertebral fractures compared with cholecalciferol and calcium therapy alone. The redistribution of bone mass from other sites to the spine has been a common concern with sodium fluoride therapy. These results also show considerable biochemical changes in markers of bone turnover and resorption in osteoporosis sufferers who may be using hormone replacement therapy, receiving only calcium citrate and cholecalciferol.
It would be worth investigating whether or not a stronger antiresorptive agent, such as a bisphosphonate or estrogen, possibly as a hormone replacement therapy, would have had more therapeutic value. The small subgroup of patients already receiving estrogen, possibly as a hormone replacement therapy, did not appear to affect response to therapy.
When parathyroid hormone (PTH) was given with estrogen in postmenopausal women with osteoporosis, urinary N-telopeptide and serum osteocalcin levels quickly increased compared with an estrogen-only group. This implies that anabolic bone agents such as fluoride and PTH could influence bone metabolism by increasing bone turnover via osteoblastic stimulation or recruitment, directly or indirectly.
These findings support the use of sustained-release sodium fluoride with calcium and cholecalciferol in treating elderly women with established osteoporosis who may have received or be receiving hormone replacement therapy. Such results suggest this combination of therapy carefully lowers the risk for vertebral fractures by stimulating new bone formation by fluoride-mediated increased osteoblastic activity. This data also reinforces earlier findings which focused on combining bone-forming and antiresorptive agents in the treatment of osteoporosis.