
Inflammation is thought to play a role in the initiation and development of cardiovascular disease, and much focus has been given to C-reactive protein (CRP), an acute-phase reactant released mainly by hepatocytes. While low-grade inflammation can predict coronary heart disease (CHD), the degree to which high CRP level is attributable to deep-rooted CHD risk factors in women using hormone therapy needs further investigation. If increased CRP level is closely linked to standard CHD risk factors, then regular screening of this biomarker would apparently be less valuable to clinicians. Thus, the study below was carried out to assess the extent to which high CRP levels may simply reflect expression of the pathobiologic changes caused by normal CHD risk factors. This study determined the use of estrogen hormone therapy in females who had surgical or natural menopause.
Findings from this analysis show that the frequency of high CRP level was higher among postmenopause women using estrogen hormone therapy than among estrogen hormone therapy nonusers or past users. Such results are in keeping with previous studies showing higher CRP levels in post-menopause women than in men and higher levels in postmenopause women undergoing estrogen hormone therapy than in postmenopause women not using hormone therapy. Yet, because of the small number of postmenopause women receiving estrogen hormone therapy, this variable was not taken into consideration in the multiple logistic regression model.
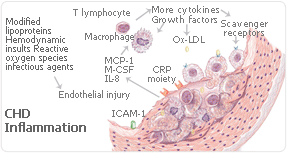
This study supports the role that traditional CHD risk factors play in atherothrombosis by showing that a key biomarker of systemic inflammation, increased CRP level, is usually linked to borderline or abnormal CHD risk factors and hardly ever/seldom occurs without them. A lot of evidence points to a strong connection between inflammatory biomarkers and CHD risk factors in post-menopause women who may be using estrogen hormone therapy.
The results outlined above indicate that high CRP level is mainly attributable to conventional CHD risk factors in women who may be using estrogen hormone therapy. Therefore, measurement of CRP levels in women who may be using or have used hormone therapy may have little clinical effect as a screening tool for CHD risk assessment unless randomized clinical trials can confirm that lowering CRP levels counteracts CHD events further than well-established lifestyle and pharmacologic modalities. Until then, greater effort should be made to promote smoking cessation, more physical activity, and lowering of elevated blood pressure, lipid levels, and visceral adiposity in women who are receiving or have received hormone therapy because all of these factors help to reduce CHD.